Introduction
Climate change affects the living world, including people, through changes in ecosystems, biodiversity, and ecosystem services. Ecosystems entail all the living things in a particular area as well as the non-living things with which they interact, such as air, soil, water, and sunlight.185 Biodiversity refers to the variety of life, including the number of species, life forms, genetic types, and habitats and biomes (which are characteristic groupings of plant and animal species found in a particular climate). Biodiversity and ecosystems produce a rich array of benefits that people depend on, including fisheries, drinking water, fertile soils for growing crops, climate regulation, inspiration, and aesthetic and cultural values.186 These benefits are called “ecosystem services” – some of which, like food, are more easily quantified than others, such as climate regulation or cultural values. Changes in many such services are often not obvious to those who depend on them.

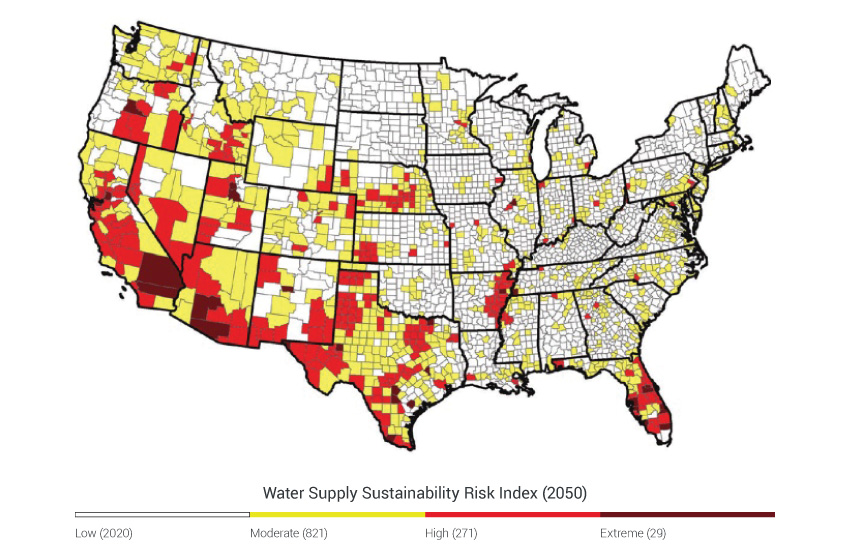
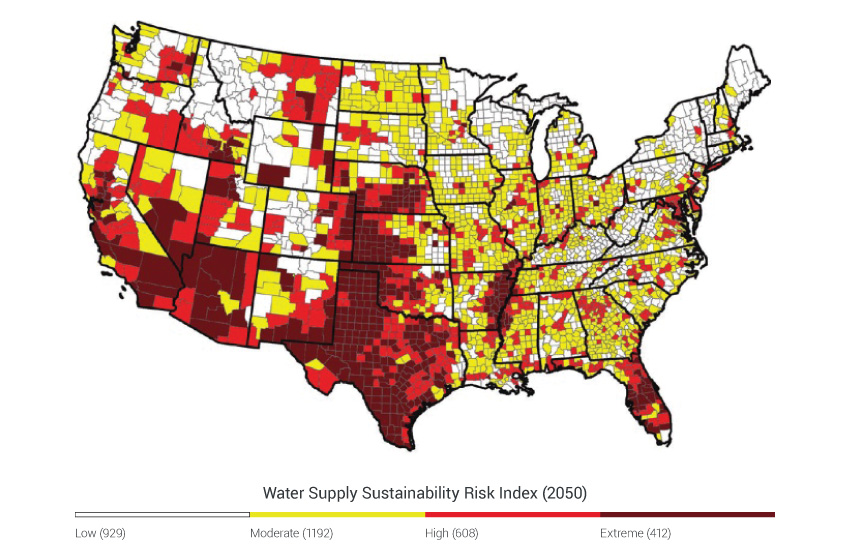
Ecosystem services contribute to jobs, economic growth, health, and human well-being. Although we interact with ecosystems and ecosystem services every day, their linkage to climate change can be elusive because they are influenced by so many additional entangled factors.187 Ecosystem perturbations driven by climate change have direct human impacts, including reduced water supply and quality, the loss of iconic species and landscapes, distorted rhythms of nature, and the potential for extreme events to overwhelm the regulating services of ecosystems. Even with these well-documented ecosystem impacts, it is often difficult to quantify human vulnerability that results from shifts in ecosystem processes and services. For example, although it is more straightforward to predict how precipitation will change water flow, it is much harder to pinpoint which farms, cities, and habitats will be at risk of running out of water, and even more difficult to say how people will be affected by the loss of a favorite fishing spot or a wildflower that no longer blooms in the region. A better understanding of how a range of ecosystem responses affects people – from altered water flows to the loss of wildflowers – will help to inform the management of ecosystems in a way that promotes resilience to climate change.